Who we are
Epidemiology Coordinating and Research (EPICORE) is a clinical trials and health
services research centre.
EPICORE is involved in the full range of activities necessary for the conduct of
clinical trials, including protocol design, case report forms design, database creation,
study management, implementation, site coordination (for multicentre trials), data
management, and analysis.
We are involved in a large number of single centre, and multicentre trials. EPICORE
is also the home of the Centre for Community Pharmacy Research and Interdisciplinary
Strategies (COMPRIS).
Mission Statement
To serve the Faculty and our community by generating new knowledge in the areas of health
and health care through the design, execution and analysis of clinical trials, health
outcomes research and epidemiologic studies.

What Investigators Are Saying About Us
“Our project application, “Nohtawiy (my father):
Supporting Cree fathers-to-be using a culturally-appropriate, community led, and
strengths-based approach: impact on maternal, infant and paternal outcomes” was
much stronger as a result of the expertise of the Alberta SPOR Support Unit and
the consultation process. Specifically, through the Consultation and Research
Services platform and face-to-face meetings we were able to develop a more
coherent and robust data capture and analysis plan, and a more meaningful
knowledge translation framework, that made sense for both our community partners
and our research group.

and Gynaecology, University of Alberta
Without exaggeration, through their support, I was able
to complete research tasks, organize a data repository, complete surveys and
help direct and support one of our graduate students. All that had previously
been hopelessly stuck or impossible to complete at my institution alone.”

University of Calgary
“The Consultation and Research Services Platform
provided support in the statistical analysis and interpretation of data from our
recent study examining a new technique in echocardiography called ‘Multi-view 3D
Fusion Echocardiography. Their contribution was integral to the success of our
research. Because of their excellent quality of work I would consider them as a
first-choice for any further research-related / statistical consultations that
our group may need to request in the future.”

Alberta
“The support and expertise of the Consultation Platform
has resulted in the long term follow-up surveys being into the REDCap platform,
including the ability for participants to provide electronic consent for
participation. The registry will now be available to all SCI patients in the
province, regardless of the etiology, or severity of their injury, time since
injury, or place of residence. The REDCap platform is very user friendly for
patients, clinicians, researchers, and community support teams.”

SCN
“Our project focused on validating the AHS foot care
tool that is used for screening feet of patient living with diabetes. The
Consultation and Research Services Platform were wonderful to work with on data
analysis for our research study. Their professional attitude and commitment to
executing the research activities allowed for quick completion of the results.?

Nutrition SCN
“The consultation services provided guidance and helped
focus the community based project I had proposed. The team was able to help me
strategize and revise the plan to meet the budget and the study objectives.
Through the networking of the team I was able to successfully run the project
through pharmacies throughout Alberta. The project was larger, more successful,
and more efficient than projects I had been involved with before.”

Alberta
“We are thankful to the SPOR Consultation and Research
Services Platform who met with us at multiple stages during the cirrhosis PRIHS
grant development phase to provide guidance around study design and methodology.
Closer to submission, Dr. Ross Tsuyuki and Dr. Yazid Al Hamarneh provided
valuable edits on the proposal which no doubt contributed to our eventual
success with our PRIHS application.”

of Alberta
“I was introduced to Ross Tsuyuki and the EPICORE
through the University Hospital Foundation, to help with development of a
clinical trial to examine the impact and effectiveness of introducing a nurse
practitioner for treatment of people with multiple sclerosis. The experience of
working with EPICORE to develop a MS clinical trial, come up with relevant
outcome measures, considering practicalities of clinical trial logistics has
been invaluable to a clinician such as myself. EPICORE has provided the support
and structure to design, then carry out a clinical trial.”

“EPICORE was responsible for designing the final CRFs,
development of the database, interacting with the sites for data submission and
entering the data into the database. To date, the recruitment closed as of
September 2016 and the CRF data is entered. The laboratory data will be
transferred over in the near future.”

Pediatrics
“I wanted to have a ‘picture’ of the pain profile of our
study participants. The EPICORE staff rose to the challenge and were able to
find the right resources. Thank you!”

Faculty of Nursing and Department of Community Health Sciences, University
of Calgary
“Our whole team appreciates the way that Dr. Tsuyuki and
his staff have
helped us through the planning, management and analytical aspects of
dealing with our precious data. They are always attentive to the nuances of
our projects and patient with the investigators, staff and students involved.
They are helping us to overcome some tremendous hurdles and make our
projects the very best.”
Agricultural, Food and Nutritional Science
“Although it took us a while to get INFORM off the
ground, working with
your team has been a pleasure – Lily in particular is responsive and on point
– I think we are going to position ourselves – with EPICORE’s help to be able
to gather and analyze this international data set in a very meaningful way.
thanks so much!”

Department of Medicine, and Associate Professor, Department of Agriculture,
Food and Nutritional Science
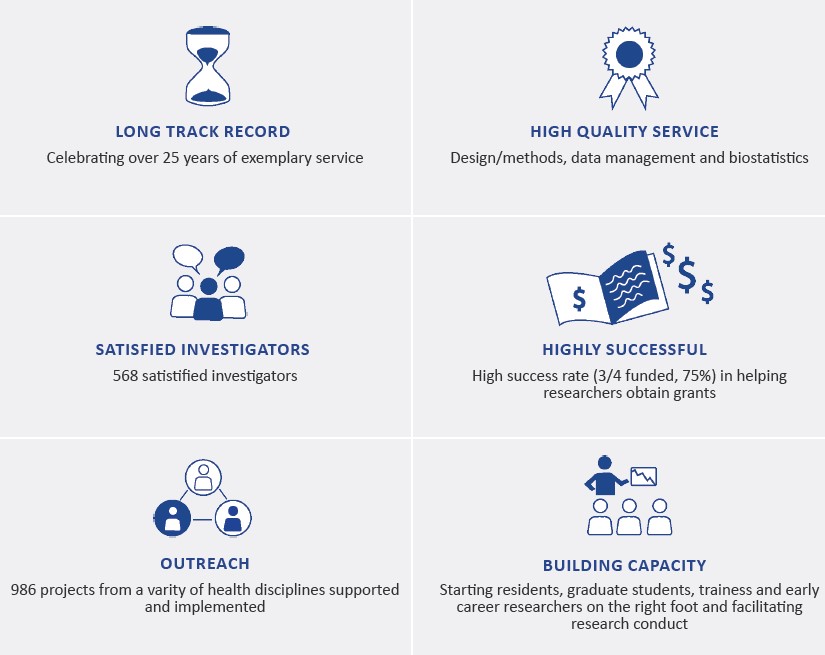
Our Services
Design/Methods
Research question refinement
Project design
Method consultation
Grant review
Data Management
Development of case report forms
Database development and maintenance
Randomization
Quality assurance
Biostatistics
Development of analytical plans
Sample size calculation
Statistical analysis
Clinical event adjudication
A ‘real life’ example of electronic integration of patient-reported outcome measures
(PROMs) into clinical care
Take some time to see why you should …
and
Interested in patient oriented research?
Book a consultation with us now
Our team











Highlighted Projects
PRxOACT Initiative
Heart disease is a common and serious medical condition which causes nearly one in every three deaths worldwide every year
Purpose: This research project aims to see whether a pharmacist-led intervention can help people reduce their risk of heart disease. The potential impact of this project is to empower people to understand how to reduce their risk of heart disease and reduce the burden of heart disease on the community […]
MAP-AP Study – Mental Health Assessment and Prescribing by
Alberta Pharmacists
MAP-AP Study – Mental Health Assessment and Prescribing by Alberta Pharmacists
Purpose: MAP-AP tracks and quantifies the impact of community pharmacists
(practicing at the full scope within Alberta) on the management of patients with
newly diagnosed Major Depressive Disorder (MDD) and/or Generalized Anxiety Disorder
(GAD), compared to usual care Setting: Community pharmacies in Calgary
Participants: […]
Images of Pharmacy Practice
Images of Pharmacy Practice Visit: Images of Pharmacy Practice website Pharmacy
practice has significantly evolved from a role focused on medication preparation and
distribution to that of an accessible, collaborative primary care practitioner.
Despite this change, many of the images of pharmacists, for example on the web
and social media, still depict a drug distribution role. […]
IGNITE
COVID-19: Bringing the ‘Invisible Layer’ of Pharmacists Professional Identity and
Roles to Light (IGNITE) Background: Pharmacists are essential frontline heroes who
have been fighting the COVID-19 pandemic since day one. They have continually
stepped up and provided the highest quality of care to their patients during
extremely challenging times. Purpose: To explore the subjectivity of
professional […]
RxPATH Study – Improving Hypertension Detection and
Control through a Hypertension Certification Program Based Upon the Hypertension
Canada Guidelines
Background/Rationale: Hypertension is the leading cause of death and disability
worldwide, accounting for more than one tenth (13%) of the total deaths every year.
Hypertension control is still elusive despite its complications (e.g., heart
disease, stroke and kidney failure) and advances in treatment. Levels of
hypertension awareness and control have increased in Canada over the […]
Outcomes of Urinary Tract Infection Management by Pharmacists:
RxOUTMAP
RxOUTMAP tracks and quantifies the impact of pharmacist management of
patients with uncomplicated urinary tract infections. Background Urinary tract
infection (UTI) is one of the most common indications for which antibacterial agents
are initiated. It is also the 8th most common reason for ambulatory clinic visits
and the 5th most common reason for emergency […]
A Quality Improvement Project to Address lower urinary tract
symptoms (LUTS) by pharmacists in the community (PILUTS)
Purpose: To determine if pharmacists in the community improve patient management of
lower urinary tract symptoms. Setting: Community pharmacies in Edmonton and
surrounding area Participants: Seniors who come to the community pharmacy Design:
randomized controlled trial Control group: Pharmacist asks some assessment questions
about LUTS Pharmacist determines the baseline scores on the patient questionnaire
Pharmacist […]
Rxcellence
Vision Our vision is to build a pharmacy network that supports the implementation of
evidence-based practice for pharmacists and technicians. Studies We Are Currently
Recruiting Pharmacists for LiveRx LiveRx is focused on equitable hepatitis C care by
pharmacists which includes: HCV screening, treatment, and curative treatments for
Albertans. LiveRx is based on partnerships between pharmacists […]
Contact Us
(HMRC) University of Alberta Edmonton, AB









